Locations
-
Stow
4277 Allen Rd
Stow, OH 44224
Call Us: (330) 928-0201
Toll Free: (800) 548-1729
Fax: (330) 926-0201 -
Kent
2013 State Rt. 59
Kent, OH 44240
Call Us: (330) 678-0201
Toll Free: (800) 548-1729
Fax: (330) 678-4272 -
Akron/Fairlawn
4099 Embassy Parkway
Akron, OH 44333
Call Us: (330) 836-8545
Toll Free: (800) 255-3671
Fax: (330) 836-8598 -
Akron (in the Valley)
(Formerly known as Kennedy Eye Center)
1558 Akron Peninsula Rd
Akron, OH 44313
Call Us: (330) 922-4040
Fax: (330) 922-1727 -
St. Clare
Surgery Center4441 Hudson Drive
Stow, OH 44224
Call Us: (330) 920-4500
Fax: (330) 920-4501 -
Medina (Reserve Commons Location)
3583 Reserve Commons Dr.
Medina, OH 44256
Call Us: (330) 722-8300
Fax: (330) 725-0445 -
Medina (Weymouth Road Location- Previously Medina Vision)
4463 Weymouth Road
Medina, OH 44256
Call Us: (330) 722-2150
Fax: (330) 722-2055 -
Wadsworth
One Park Centre Dr.
Suite 106
Wadsworth, OH 44281
Call Us: (330) 334-1300
Fax: (330) 334-5300 -
Downtown Canton
Previously Eye Centers of Ohio
800 McKinley Ave NW
Canton, OH 44703
Call Us: (330) 452-8884
Fax: (330) 452-2404 -
Institute for Refractive & Intraocular Surgery (IRIS)
800 McKinley Ave NW
Canton, OH 44703
Call Us: (330) 639-0046
Fax: (330) 639-0042 -
North Canton
Previously Eye Centers of Ohio
6407 Frank Ave NW
North Canton, Ohio 44720
Call Us: (330)966-1111
Fax: (330) 966-8333 -
Streetsboro
(Formerly known as Clear Vision Centers)
1155 Oh-303
Streetsboro, OH 44241
Call Us: (330) 422-2020
Fax: (330) 422-0316


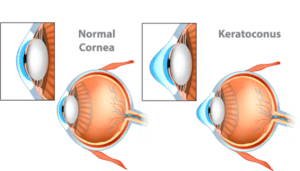
 The physicians at the Cornea Center at Northeast Ohio Eye Surgeons offer the most technologically and surgically advanced care, delivered with kindness and compassion.
The physicians at the Cornea Center at Northeast Ohio Eye Surgeons offer the most technologically and surgically advanced care, delivered with kindness and compassion. The cornea is the clear, dome-shaped window that covers the front of the eye. It is similar to the glass face of a watch or the outermost glass lens in a camera.
The cornea is the clear, dome-shaped window that covers the front of the eye. It is similar to the glass face of a watch or the outermost glass lens in a camera.